The origin of life on Earth is still enigmatic, but we are slowly unravelling the steps involved and the necessary ingredients. Scientists believe life arose in a primordial soup of organic chemicals and biomolecules on the early Earth, eventually leading to actual organisms.
It’s long been suspected that some of these ingredients may have been delivered from space. Now a new study, published in Science Advances, shows that a special group of molecules, known as peptides, can form more easily under the conditions of space than those found on Earth. That means they could have been delivered to the early Earth by meteorites or comets—and that life may be able to form elsewhere, too.
The functions of life are upheld in our cells (and those of all living beings) by large, complex carbon-based (organic) molecules called proteins. How to make the large variety of proteins we need to stay alive is encoded in our DNA, which is itself a large and complex organic molecule.
However, these complex molecules are assembled from a variety of small and simple molecules such as amino acids—the so-called building blocks of life.
To explain the origin of life, we need to understand how and where these building blocks form and under what conditions they spontaneously assemble themselves into more complex structures. Finally we need to understand the step that enables them to become a confined, self-replicating system—a living organism.
This latest study sheds light on how some of these building blocks might have formed and assembled, and how they ended up on Earth.
Steps to Life
DNA is made up of about 20 different amino acids. Like letters of the alphabet, these are arranged in DNA’s double helix structure in different combinations to encrypt our genetic code.
Peptides are also an assemblage of amino acids in a chain-like structure. Peptides can be made up of as little as two amino acids, but also range to hundreds of amino acids.
The assemblage of amino acids into peptides is an important step because peptides provide functions such as “catalyzing,” or enhancing, reactions that are important to maintaining life. They are also candidate molecules that could have been further assembled into early versions of membranes, confining functional molecules in cell-like structures.
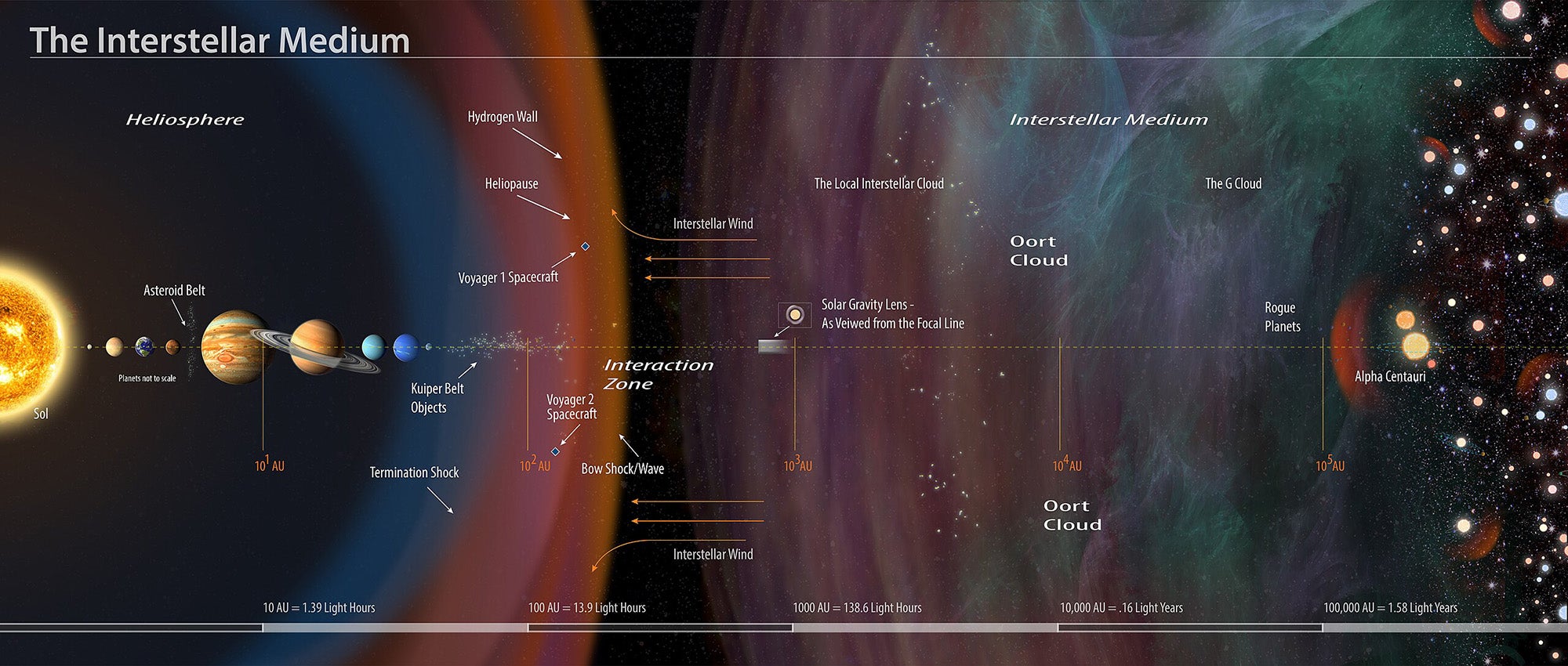
However, despite their potentially important role in the origin of life, it was not so straightforward for peptides to form spontaneously under the environmental conditions on the early Earth. In fact, the scientists behind the current study had previously shown that the cold conditions of space are actually more favorable to the formation of peptides.
In the very low density of clouds of molecules and dust particles in a part of space called the interstellar medium (see above), single atoms of carbon can stick to the surface of dust grains together with carbon monoxide and ammonia molecules. They then react to form amino acid-like molecules. When such a cloud becomes denser and dust particles also start to stick together, these molecules can assemble into peptides.
In their new study, the scientists look at the dense environment of dusty disks, from which a new solar system with a star and planets emerges eventually. Such disks form when clouds suddenly collapse under the force of gravity. In this environment, water molecules are much more prevalent—forming ice on the surface of any growing agglomerates of particles that could inhibit the reactions that form peptides.
By emulating the reactions likely to occur in the interstellar medium in the laboratory, the study shows that, although the formation of peptides is slightly diminished, it is not prevented. Instead, as rocks and dust combine to form larger bodies such as asteroids and comets, these bodies heat up and allow for liquids to form. This boosts peptide formation in these liquids, and there’s a natural selection of further reactions resulting in even more complex organic molecules. These processes would have occurred during the formation of our own Solar System.
Many of the building blocks of life such as amino acids, lipids, and sugars can form in the space environment. Many have been detected in meteorites.
Because peptide formation is more efficient in space than on Earth, and because they can accumulate in comets, their impacts on the early Earth might have delivered loads that boosted the steps towards the origin of life on Earth.
Weekly Newsletter
So what does all this mean for our chances of finding alien life? Well, the building blocks for life are available throughout the universe. How specific the conditions need to be to enable them to self-assemble into living organisms is still an open question. Once we know that, we’ll have a good idea of how widespread, or not, life might be.![]()
This article is republished from The Conversation under a Creative Commons license. Read the original article.








